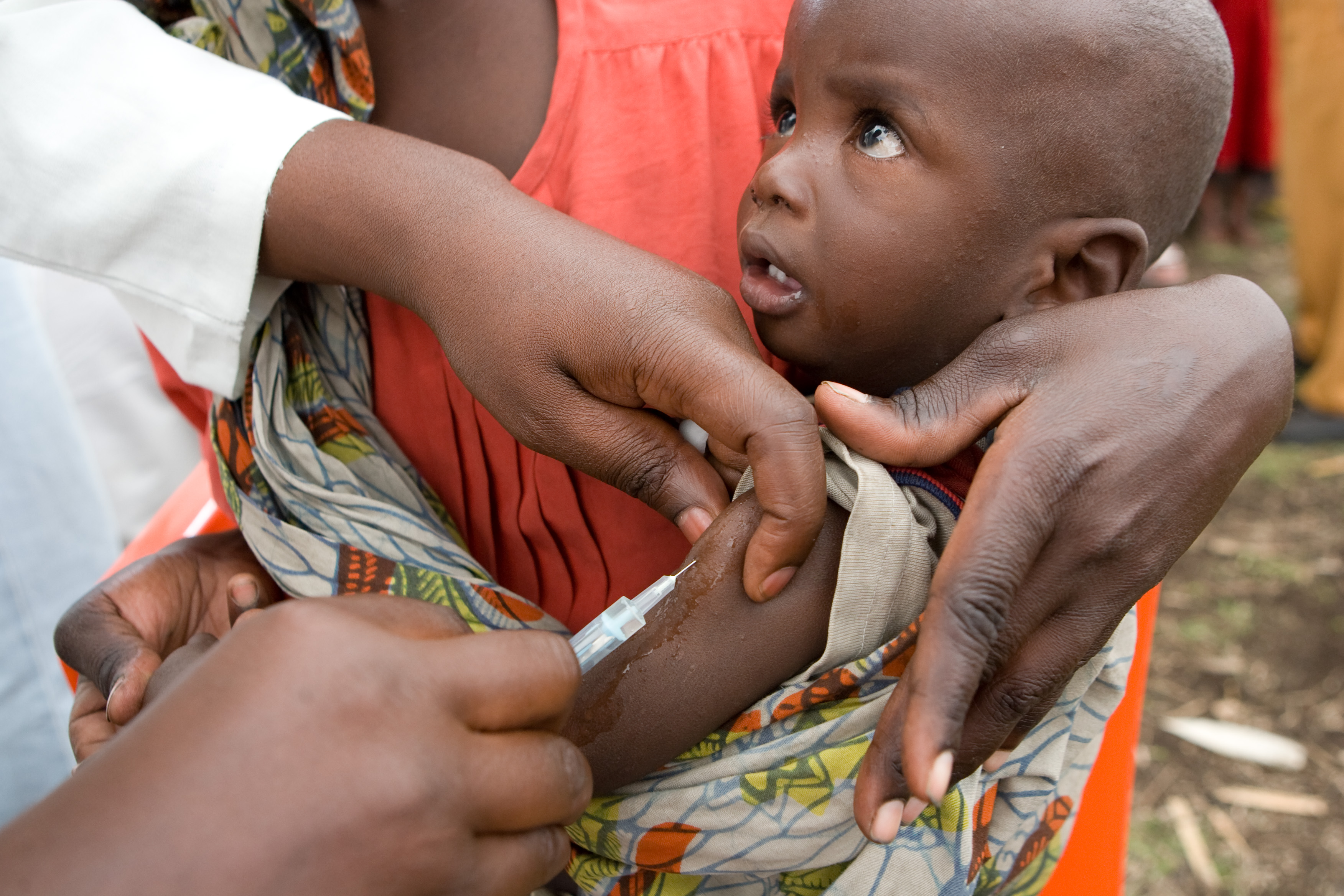
For decades, there has been consistent chatter, research, and hope for a potential malaria vaccine. Now, all three are finally coming to fruition to roll out the world’s first clinical malaria vaccine trials. The World Health Organization Regional Office for Africa (WHO/AFRO) announced today that Ghana, Kenya, and Malawi have been chosen for the WHO-coordinated pilot implementation program that will make the world’s first malaria vaccine available in 2018.
“The prospect of a malaria vaccine is great news. Information gathered in the pilot will help us make decisions on the wider use of this vaccine,” said Dr Matshidiso Moeti, WHO Regional Director for Africa, in a statement. “Combined with existing malaria interventions, such a vaccine would have the potential to save tens of thousands of lives in Africa,” she added.
Sub-Saharan Africa records 90% of all global malaria cases. Even though the number of cases and deaths have dropped dramatically since 2001, the rate is still astronomically high. In fact, malaria still remains one of the deadliest killers on the African continent, especially for children under the age of five.
To date, the most effective way to curb malaria cases is via the use of bed nets and indoor residual spraying. Unfortunately, 43% of sub-Saharan Africans are not protected against either and 429,000 people died from malaria in 2015. After spending time with mothers in Tanzania with Malaria No More, I saw this to be true. I met moms standing in long lines to receive new nets, but the ones they had used for years had holes throughout, rendering them virtually worthless.
There is now new hope to curb child deaths with the injectible malaria vaccine targeted to children within five to 17 months called RTS,S. The vaccine developed by GlaxoSmithKline. Malawi, Kenya, and Ghana were chosen for the following reasons according to the World Health Organization:
- high coverage of long-lasting insecticidal treated nets (LLINs)
- well-functioning malaria and immunization programs
- a high malaria burden even after scale-up of LLINs,
- and participation in the Phase III RTS,S malaria vaccine trial
The countries themselves will determine the areas in their country where the trials will ultimately take place. The $49.2 million cost of the trials will be taken up by Gavi, the Vaccine Alliance, the Global Fund to Fight AIDS, Tuberculosis and Malaria, and UNITAID. The World Health Organization and GlaxoSmithKline will additionally provide complimentary funds for the malaria trial efforts.
Photo: UN Photo/Marie Frechon






Leave a Reply